
 |
||
DMSO Background Literature |
|||||||||||||||
Further
Observations on the Effect of Dimethyl Sulfoxide in
Patients with Generalized Scleroderma (Progressive Systemic
Sclerosis)
Arthur L. Scherbel, Lawrence J. McCormack, and John K. Layle Departments of Rheumatic Diseases and Pathology, Cleveland Clinic Foundation Cleveland, Ohio
Systemic scleroderma (progressive systemic sclerosis) is a connective tissue disorder of unknown etiology. According to Talbott,1 the disease is ultimately fatal, although duration of life following onset of symptoms is extremely variable and unpredictable in most patients. Varying degrees of cutaneous and subcutaneous involvement of the hands, forearms or face are generally observed. Raynaud's phenomenon occurs almost consistently and subsequently, ischemic ulcers may appear at the fingertips, elbows or ankles. As the disease progresses, all systems of the body may be affected. Eventually, disabling changes in skin, muscles and joints may be associated with circumscribed or interstitial calcinosis. Pathologic changes involving the connective tissue have not been clearly defined and opinions vary regarding the abnormal changes that occur. Some investigators report that there are no abnormalities in the collagen or amorphous ground substance. 2, 3 Others believe that amorphous material is increased,4, 5 while still others say that the collagen fibers are increased. 6 Various other studies have suggested that there are biochemical abnormalities within the connective tissue, i.e., depolymerized ground substance7 and increased cutaneous hexosamine. 8 Numerous therapeutic measures, including corticosteroids, sodium endrate, reserpine, serotonin inhibitors, potassium aminobenzoate, chloroquine, and cytotoxins have been used, but none is consistently or highly effective. The dismal outlook regarding treatment of this disease is reflected in an editorial comment in the Sixteenth Rheumatism Review, which states, "We doubt the efficacy of any therapy for systemic sclerosis."9
In April, 1965, Scherbel et al. published a preliminary report10 on the effect of dimethyl sulfoxide (DMSO) in ten patients with progressive systemic sclerosis. The study has been continued and now includes 42 patients (27 women and 15 men), aged 20 to 69 years, who have been treated with DMSO for periods ranging from three to twenty-three months. Duration of the disease in these patients ranged from 1-25 years. All patients had varying degrees of systemic involvement. At the time treatment was started, the disease was considered mild in 18, moderate in 19, and severe in five patients, according to the criteria (Table 1) we have established for grading the severity of the disease. Cutaneous manifestations were present in all patients treated; nineteen of them had ischemic ulcers of the finger tips. Raynaud's phenomenon was observed in all but one patient. Gastrointestinal involvement included delayed esophageal peristalsis, esophagitis with and without obstruction, esophagitis with bleeding, diminished gastric peristalsis, delayed intestinal peristalsis, ileus with abdominal distention simulating intestinal obstruction and malabsorption, and was present in 25 patients. Pulmonary involvement in 21 patients included roentgenographic changes, chronic nonproductive cough, pulmonary infection and impaired ventilatory function. Fifteen patients had cardiac symptoms which included dyspnea, orthopnea, palpitation, tachycardia, cardiac enlargement, gallop rhythm, and edema. Hypertension manifested by persistent elevation of blood pressure over 150/110 was present in five patients. Renal impairment was observed in four patients. In each case, there was elevation in concentration of blood urea nitrogen and increased urinary sediment in the 12-hour Addis count.
Criteria for Assessing Severity of Progressive Systemic Sclerosis
Each patient served as his own control inasmuch as previous treatment had been ineffective. Laboratory studies, including hemoglobin, leukocyte count, differential leukocyte count, serum electrophoresis, serum glycoproteins (Shetlar),11 roetgenogram of the chest, esophagram, electrocardiogram, cutaneous biopsy and 24-hour urinary hydroxyproline output were obtained on 21 hospitalized patients before treatment with DMSO was started. Biopsies of the skin and 24-hour urinary hydroxyproline determinations were repeated at intervals of six to eight weeks in 12 patients, and of twelve to sixteen weeks in eight patients. Serial roentgenograms of the hand, chest and esophagus were obtained every three to six months. Patients without extensive internal sclerodermatous changes received out-patient treatment and returned for reevaluation at eight-week intervals. Blood studies, hemoglobin, leukocyte and differential counts, serum transaminase, and alkaline phosphatase determinations were obtained at each visit. Other tests carried out to aid in evaluation of skin changes at each return visit included grip strength, ability to flex fingers, reactions to intradermal serotonin, comparison of frequency of vasomotor color changes (Raynaud's phenomenon) that occurred daily and examination of ischemic ulcers of the finger tips. Methods of Administration of Dimethyl Sulfoxide Initially, the chemical was applied topically to affected areas in concentrations ranging from 30% to 60%. Subsequently, the concentration was increased to 70% and, in some cases, to 100%. It was soon observed that most patients did not tolerate the same concentration of DMSO over all areas of the body and that the rapidity of cutaneous absorption varied considerably in different regions. The neck, face, and axillas were more sensitive than the back or extremities. Twelve patients received topical DMSO to the entire body area in concentrations ranging from 30% to 100%, depending upon the area of the body painted and the duration of treatment. After DMSO was applied, it was allowed to penetrate the skin until absorption was complete (approximately 30 min). After periods of two to three weeks, application was limited to the hands, forearms, feet and, occasionally, the face. Usually, the solvent was applied three times daily for approximately three months, then twice daily, until softening of the skin and the range of motion were nearly normal, and then reduced to one application daily. The total dose of DMSO applied to the entire body was estimated at 60 to 100 gm daily for periods ranging from two to three weeks. The total daily dose for application to the extremities was estimated to be 50 to 75 gm. In 12 patients, DMSO was applied only to the extremities, and after three months, these patients applied it only to the forearms and hands (estimated dose 25 to 30 gm daily). Eighteen patients in the entire group have continued to apply DMSO in this manner for periods ranging from 10-14 months. The total dose of DMSO during this time is estimated to range from 10.8 kg to 12.6 kg. It has not been possible to determine how much of the drug applied to the skin was actually absorbed into the body. In most cases, a burning or sharp prickling sensation associated with transient erythema occurred during the first few applications of DMSO. In certain patients, there was temporary blistering of the skin, prolonged severe itching, or hives, suggesting histamine release, especially when the initial concentration was excessive during the first few days of therapy. Cutaneous reactions usually subsided in the majority of patients after seven to ten days, at which time the concentration of DMSO could be increased without recurrence of cutaneous reactions. Immersion of the hands and wrists in DMSO was used in ten patients who did not respond satisfactorily to topical application of the chemical for three months. Ten patients were treated solely by immersion of the hands and/or feet. The hands were usually immersed for one min in 50% DMSO. The time was increased daily until the period of immersion was five to ten minute twice daily. In all cases, those patients who responded unsatisfactorily to topical application of DMSO responded satisfactorily to immersion of the hands. Usually, the concentration was not increased above 70% and, in many patients, it was maintained at 50%; this resulted in less drying of the skin. The dilute solution of DMSO used for immersion was poured back into a separate bottle after completion of each soaking and was discarded after it had been used for one week. Nine patients received DMSO subcutaneously in amounts ranging from 5 to 10 ml of 1% to 5% concentration, diluted in physiologic saline solution, because certain lesions, e.g., circumscribed and interstitial calcinosis, tendon contractures, and capsular adhesions treated with topical dimethyl oulfoxide, did not improve satisfactorily. Injections were made at daily intervals for four days each week for two to four weeks in one to ten injection aites. The total dose of DMSO administered subcutaneously ranged from 0.1 to 5 gm daily.
Evaluation of treatment in 42 patients with systemic sclerosis confims our initial impression10 that cutaneous manifestations are slowly but definitely alleviated in certain patients by application of DMSO. In most instances, long-term therapy of one to two years is necessary to obtain significant cutaneous improvement. The initial burning and itching that occur in the majority of patients usually subside spontaneously after one or two weeks of treatment. These skin reactions can be minimized by applying DMSO in concentratiorm of 35% to 50% during the first two weeks and then gradually increasing the concentration to 75%. If the immersion technique is used, the concentration need not exceed 75%; in most instances, a 50% concentration is sufficient. As treatment progresses, the skin usually becomes dry, coarse and furrowed. In certain patients with extensive skin involvement, the skin may appear nodular. As joint motion and skin pliability returned toward normal dimethyl sulfoxide was stopped on one hand and continued on the other in three patients. Much to our surprise the skin returned to normal appearance and texture over the hand no longer receiving treatment in four to six weeks in each case. This unusual response has been associated with normal appearance of collagen in these areas (Figure 1a & 1b). In order to each patient as objectively as possible, certain criteria have been used for grading the amount of cutaneous improvement (Table 2). These criteria prove helpful if doubt exists regarding the efficacy of treatment. Control studies were carried out in certain patients by treating only one extremity and comparing it with the other. According to these criteria for evaluation of cutaneous improvement in 42 patients, 26 showed good or excellent improvement and 16 showed fair or poor response to therapy. Of the 26 with good to excellent response, three have discontinued therapy after one year because skin changes, range of joint motion, grip strength, and histologic changes have returned to normal. In each of these cases, improvement has been maintained six months after cessation of treatment. It is our opinion that they have gone into a spontaneous remission while undergoing treatment. The other 23 patients are maintaining good to excellent response while continuing to use DMSO, mainly in the form of hand soaks and topical application to the forearms. Treatment was discontinued temporarily in nine of these patients because of satiafactory improvement, but, in each instance, pain and stiffness returned within four weeks, and then treatment was resumed.
Criteria for Cutaneous Improvement in Progressive Systemic Sclerosis
Ischemic Ulcers Ischemic ulcers were present in 19 of the 42 patients. In 12, treated with topical DMSO, the ulcers healed within one to three weeks in six patients and within three to six weeks in three. Ulcers failed to heal in three patients treated with topical applications of DMSO. One of these discontinued treatment because of increased pain and the other two patients were treated by immersion of the finger tips in 50% DMSO solution. In both instances, the ulcers healed within one week. Thereafter, immersion of the hands was substituted for topical application for ischemic ulcers and also for patients who failed to show satisfactory improvement in cutaneous manifestations with topical application of the chemical. Since the immersion technique has been substituted for topical application, no resistant ischemic ulcers have been encountered. Cutaneous improvement in the fingers has also been more rapid with this method of therapy. (Figures 2a & 2b) Circumscribed and Interstitial Calcinosis In two patients undergoing treatment with DMSO, circumscribed and interstitial calcinosis has decreased (Figures 3a & b). A young woman aged 19 years, had severe fixed flexion contractures involving both elbows and knees. In the right elbow, there was a fixed flexion contracture at 30°; in the left elbow, motion was restricted to 30° to 90°. Injection of 10 ml of 5% DMSO into the involved site daily for four days a week for four weeks resulted in marked disappearance of interstitial and circumscribed calcium deposits and return of joint motion, after three months, from 30° to 170° in the right elbow. Range of motion in the left elbow increased from 30° to 90° to 30° to 180°. The knees, which had had fixed flexion at 90°, had a range of motion of 45° to 175°. Roentgenograms of the right lower extremity showed marked loss of interstitial calcium while receiving DMSO treatment. During this time the serum calcium increased from 10.5 mg/100 ml to 13.5 mg/100 ml for approximately six weeks, after which time it returned to normal, without alteration in DMSO therapy.
As this study continues, it is apparent that certain patients with systemic sclerosis respond to DMSO therapy more readily and completely than others, while some either do not improve, or may relapse into a rapidly downhill course. With use of our criteria for evaluating cutaneous improvement and comparison of the response with the severity of the disease in each patient before treatment (Tables 1 and 2), it becomes apparent that patients with relatively mild disease are most likely to respond more favorably to DMSO than those with severe or advanced disease. Sixteen of 26 patients who have shown good or excellent response to treatment were classified as having minimal disease; eight had moderately severe disease, and only two had severe or far advanced disease. Six patients with advanced progressive disease either died during treatment or within three months after treatment with DMSO. There was no evidence of a causal relationship to therapy, although the possibility that this treatment might aggravate renal impairment in seriously ill patients with preexisting kidney disease cannot be completely excluded. Side Effects and Manifestations of Toxicity No serious side effects or toxicity manifestations were encountered while treating this group of patients. Ophthalmologic examination in 22 patients failed to reveal any lesions that could be related to DMSO. The side effects most frequently observed in patients undergoing treatment with DMSO were skin irritation and the characteristic "garlic" breath odor. All patients noted a burning or prickling sensation of the skin area to which the solvent was applied. Response of the patients to this irritation varied greatly. Most patients were willing to continue treatment when they were told the irritation would subside and a few patients reported this reaction only after they were questioned specifically about burning and itching of the skin. An interesting clinical observation that has emerged from this study is that persistent itching of the skin occurs much more commonly in patients with progressive systemic sclerosis than we had previously realized. Eleven patients stated that generalized itching was a constant annoyance accompanying their disease and often prevented sound sleep; five of them had excoriations of their skin when they were first examined. Four patients noted transient angioneurotic edema associated with itching and erythema immediately following topical application of 80% DMSO. In two patients, this reaction persisted for 24 hours. After the chemical was diluted to 50% concentration, the skin reaction disappeared and did return following gradual increase in DMSO concentration to 80% after one month of treatment. This reaction was thought to be related to histamine release. A garlic-like breath odor was noted by all patients and was more evident during the first month of treatment. After smaller areas of the body were treated, the odor was less noticeable. Only one patient discontinued treatment because of this annoyance. Lethargy, generalized fatigability, and increased muscular weakness were prominent complaints before treatment in 27 patients. Fifteen showed good improvement with respect to these symptoms, subjectively, and in increased grip strength. Twelve patients complained of increasing muscular weakness after starting DMSO therapy. In five of these, there was some improvement when the concentration applied was reduced to 50%. In two of these patients who complained of aggravation of weakness while receiving DMSO, interruption of treatment apparently resulted in increased strength. Before starting DMSO, one of these last two patients, who had progressive systemic sclerosis and polymyositis, was not doing well with a treatment of 7.5 mg of prednisone and 10 mg of isocarboxyzide (Marplan) daily. After starting topical applications of DMSO, she complained of increasing muscular weakness. After three months, DMSO was discontinued and the previous therapy was reinstated. Much to our surprise, muscular strength began to improve almost immediately after DMSO was withdrawn and has contined to improve progressively each month. Three months after stopping DMSO, the patient stated that she was stronger than she has been at any time since she became 11 five years previously and significantly stronger than she was before starting treatment with DMSO. In this case, grip strengths have increased 50% bilaterally. Anorexia or nausea or both occurred in eight patients and was associated with loss of weight of 3-10 lbs. These symptoms disappeared in five patients when the dose of DMSO was reduced. Elevation in levels of serum glutamic oxaloacetic transaminase (SGOT) was observed in eight patients. Three patients had increased SGOT before reatment with DMSO was started, --110, 90, and 41 U, respectively. All three evels dropped to normal during the first two months of treatment. The other five patients had normal levels of SGOT (below 40 units) before treatment was started. Three of the five had levels of 40-57 U one month, three months and six months, respectively, after DMSO was started. All three patients have continued to soak their hands in 50% DMSO for five minutes twice daily. After two months, the SGOT levels again decreased to normal and have remained so. The elevation in transaminase was not accompanied by any abnormality in other liver function tests. It is thought that this change might have reflected an inflammatory reaction in muscles. Alterations in Urinary Hydroxyproline Hydroxyproline occurs in the body almost exclusively in collagen, where it accounts for 13% of the total amino acids,12 It is excreted in the urine in bound and free forms. 13, 14 Bound hydroxyproline, presumably in a polypeptide, is released by acid hydrolysis and accounts for nearly all the urinary excretion of hydroxyproline. Because almost all hydroxyproline is present in collagen, it has been postulated that, if there were a significant deviation from normal in the metabolism of collagen or an increase in degradation of this protein in patients with collagen diseases, these changes might be reflected by increased levels of excretion of this amino acid in the urine. Ziff14 found no significant difference in excretion of total hydroxyproline in normal adulft and adults with rheumatoid arthritis and collagen diseases, including two with progressive systemic sclerosis. This suggests either that collagen is not broken down in these diseases or that, if there is collagen degeneration, it is not sufficient to increase the excretion of hydroxyproline. Increased hydroxyproline excretion associated with abnormal bone metabolism, as in Paget's disease and hyperparathyroidism may reflect turnover in collagen of bone. 12 Total 24-hour urinary excretion of hydroxyproline was determined in 21 patients with progressive systemic sclerosis before treatment with DMSO was started. Pretreatment values ranged from 20-60 mg. Rapid increases in 24-hour urinary levels occurred during the first week of treatment in six of the 21 patients. Average increase of urinary hydroxyproline during the first month of treatment was greater than 50% above the average pretreatment urinary output. This increase usually persisted for two to three months and then gradually fell to pretreatment levels. Figure 4 shows the transient increase in three patients who had serial 24-hour hydroxyproline determinations at monthly intervals for six months or longer.
Average 24 Hour Urinary Hydroxyproline in 3 Patients 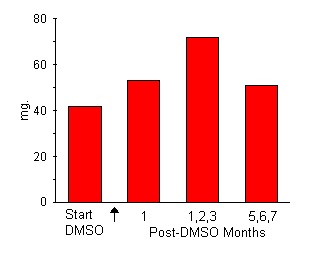
Figure 4: Average Excretion of hydroxyproline in urine (24 hours) in three patients during treatment with DMSO.
After preliminary experimentation with various staining methods, the battery used for histologic study of the sections of sclerodermatous skin includes hematoxylin and eosin, the Armed Forces Institute of Pathology modification of Mowry's acid mucopolysaccharide stain (using Müller's colloidal iron) before and after buffered hyaluronidase digestion, and the Gömöri elastic stain. Nine patients treated early in the series have been studied with a sequence of biopsies. Four have had two; one, three; one, four; and three have had five spaced biopsies. The time interval from onset of treatment to appearance of morphologic changes varied from one to seven months. The major change noted in biopsies from sclerodermatous areas while under treatment with DMSO was the development of pronounced staining of the entire collagen area with the colloidal iron (Mowry's acid mucopolysaccharide stain). The area immediately adjacent to the epidermis took on a brilliant blue color, and there was some interdigitation of the blue coloration with apparently mature collagen in the dermis, which is characteristic of progressive systemic sclerosis. It seemed to follow where the collagen bundles had been. This material digested readily with hyaluronidase, so the blue staining was quenched in the digested sections. The pretreatment controls, on the other hand, contained only sparse amounts of acid mucopolysaccharide, even less than that in normal skin; the material was found around sweat glands, and occasionally in mute patches throughout the collagen. The elastic tissue stains in treated sclerodermatous areas uniformly showed excellent retention of elastic fibers throughout the area of the original dermis (Figure 5). In one patient who had serial biopsies, the reaction for acid mucopolysaccharide disappeared three months after beginning DMSO treatment and, in another, six weeks after cessation of treatment. When this patient resumed treatment six months later, the reaction reappeared. Another patient had a most interesting sequence of events in that the reaction was positive six weeks after she started topical treatment and then disappeared in the area of treatment, which showed no clinical improvement. Then, after four months of additional treatment by soaking, the biopsy was again positive. The most interesting case of all, however, was one in which the changes were never pronounced in three biopsies over a period of eight months. All sections showed some acid mucopolysaccharide that had digested, but not in major quantities as compared with other cases. At the end of sixteen months of treatment, a most amazing zone of collagen appeared in the upper dermia between the epidermis and the underlying sclerodermal type of collagen. This layer has all the appearance of normal wavy collagen and contains embedded normal elastic fibers. This correlates very closely with the clinical change to skin that appears normal (Figures 6a & 6b).
The effect of DMSO on sclerodermal tissue is speculative, but it appears that after the diseased skin has been treated with this material, major histochemical alterations can be demonstrated in the form of increased amounts of acid mucopolysaccharide within the involved and treated area. It is believed that this material, which is digested by hyaluronidase, is actually acid mucopolysaccharide. It is justifiable to presume that this must be derived from the collagen in the area. Therefore, it would seem that collagen is being broken down and returning to the mucopolysaccharide moiety phase. In one case, there is histologic evidence that prolonged continuation of this process can lead to new formation of normal-appearing dermis.
The most dramatic finding in this study of 42 patients with scleroderma treated with DMSO is the evidence of histochemical changes that may occur with prolonged treatment. This process needs further study in a larger series of cases to determine the exact mechanism of this reaction. At any rate, this histochemical change appears to correlate with the cutaneous changes observed clinically. The healing of ischemic ulcers of the finger tips has been consistently observed. This probably can be explained by increased nutrition resulting from relief of vascular spasms. In 50% of patients with Raynaud's phenomenon, this has disappeared completely, and all patients have been significantly improved as shown by the decrease of color change. The mechanism of action accounting for disappearance of localized and interstitial calcinosis, with consequent increase in range of motion, is not known. The relief of pain, stiffness and, to a lesser extent, weakness in these patients who have been treated with DMSO is impressive, and most of the patients are continuing maintenance treatment. It appears that DMSO, as administered at present, has no effect on the internal manifestations of scleroderma. Of 26 patients who had good to excellent improvement, 16 had minimal, and eight had moderate disease, according to our criteria for classification of the severity. It should be emphasized, however, that the results seen in treating patients with scleroderma with DMSO have never been observed with any other method of therapy. It is the first time we have observed evidence, both clinical and histological, that the collagen is undergoing definite change.
aDimethyl sulfoxide used in this study was supplied through the courtesy of Dr. Richard Brobyn, Associate Medical Director, Merck, Sharp & Dohme, West Point, PA.
Annals New York Academy of Sciences, 1968, pp. 613-629. DMSO Organization wishes to thank the publishers of Annals New York Academy of Sciences for allowing this article to be placed on our World Wide Web site. The publisher retains all copyright. Please obtain permission from the publisher for permission to copy or reprint any portion of this article. |
|||||||||||||||
© 2001-2022
All rights reserved |